|
Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen). Electrons can only be in certain energy levels, and they must gain energy to move to a higher level, and must lose energy to go to a lower level. The Law of Definite Proportions applies when elements are reacted together to form the same product. Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. Dalton himself told Thomas Thomson in 1804 that he had been. atoms of an element are identical to one another but different from atoms of other elements. atoms of one element cannot be converted into atoms of another element. 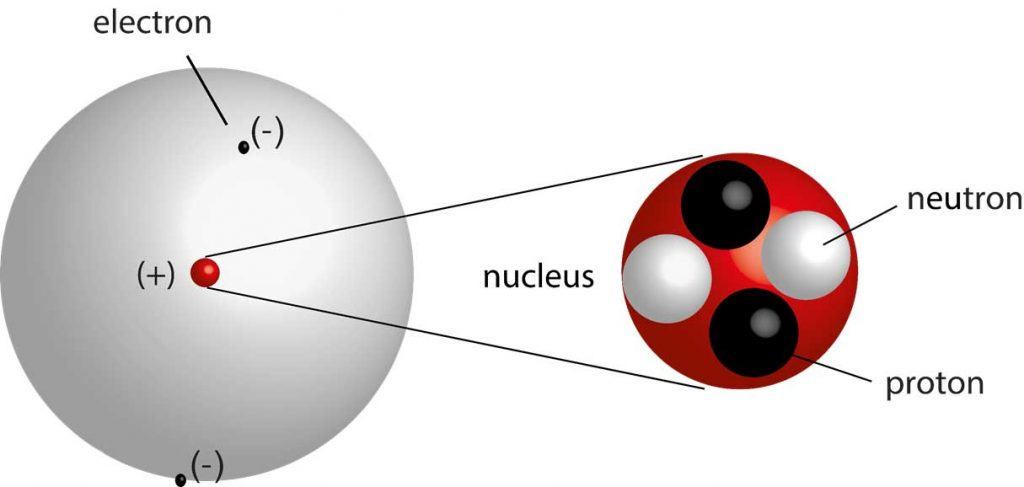
Electromagnetic Spectrum, Reflection, Refraction, Light and Lenses, Light and Colour, The Eye. Chemistry Test 2- The Modern Atomic Theory 3. Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. With regard to the genesis of the theory in his own mind much doubt has prevailed until recently. Daltons 'modern' atomic theory, in todays terms, would be something like this: all matter consists of indivisible atoms which cannot be created or destroyed. The Modern Atomic Theory Assignment and Quiz. If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C. 
\): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B. The Theory of Spectra and Atomic Constitution: Three Essays.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
